23 Jan Does Peel-Packing Put You at IFU Compliance Risk?

Common Strategies to track reprocessing cycles may sound good in theory, but day-to-day reality exposes significant flaws.
If you work in a hospital that performs robotic surgery with Intuitive Surgical’s da Vinci® Xi®, Si®, or SP® robots, then you may know that their instruments don’t last forever. In their IFUs, Intuitive Surgical specifies a maximum for both surgical uses (surgical procedures performed) and reprocessing cycles for each da Vinci instrument. To stay compliant with da Vinci’s IFU standards, hospitals are required to have a method to determine when their da Vinci instruments have reached their specified limit for both surgical uses and reprocessing cycles in order to know when to remove them from service. Removing “expired” instruments from service is extremely important for patient safety and IFU compliance.
What da Vinci Offers:

Fortunately, the da Vinci system provides a built-in method to track surgical uses— If an instrument is attached to the da Vinci robot, the system will indicate if it has reached the maximum number of surgical uses. Additionally, each instrument has an internal mechanism that provides you with a visual indicator when the surgical use maximum has been reached. However, the robot solves only one of the two tracking requirements, as hospitals still must track reprocessing cycles in order to stay compliant with IFU standards.
The Issues with Peel-Packing:
A common approach to tracking the reprocessing cycles of da Vinci instruments involves “peel-packing”— an easy, but risky solution. This method calls for placing a single da Vinci instrument into a peel pouch (or wrap) before the sterilization process and only opening the instrument once it is time to be used by the robot.
The peel-packing approach poses two risks:
- Cautery cord surgical uses and reprocessing cycles could exceed the specified number of uses, as the robot doesn’t track these accessories.
- All da Vinci instruments could exceed the maximum reprocessing cycles, as surgical uses can easily become misaligned with the true number of reprocessing cycles for a da Vinci instrument.
Let’s dig in a little deeper.
Peel-Packing: The Assumptions and Reasonings Behind the Approach
The assumptions and reasoning behind the “peel-packing” approach are:
- The maximum reprocessing cycles for a specific instrument are always greater than the maximum surgical uses.
- The da Vinci system will alert the user once the instrument has reached its surgical-use maximum, and the instrument has a visual indicator (“remove from service” alert).
- When the instrument is used in a da Vinci procedure, the robot will track the surgical uses.
- Surgical uses and reprocessing cycles will always be equal because a pouch won’t ever be opened unless the instrument is used in a da Vinci procedure. Therefore, surgical uses are a valid proxy for reprocessing cycles.
If all of these assumptions were always true, a da Vinci instrument could never (theoretically) exceed the reprocessing cycle maximum. But, some of these assumptions are not always true. Let’s take a look at them one by one.
We will begin with the easy ones first:
Assumption #1 is (currently) true. As of the publish date for this blog, all of the da Vinci instruments have maximum reprocessing cycles exceeding their maximum surgical uses. Additionally, assumption #2 is (currently) true — the da Vinci system provides an alert, and the instrument provides a visual indicator when the instrument has reached its maximum surgical uses.
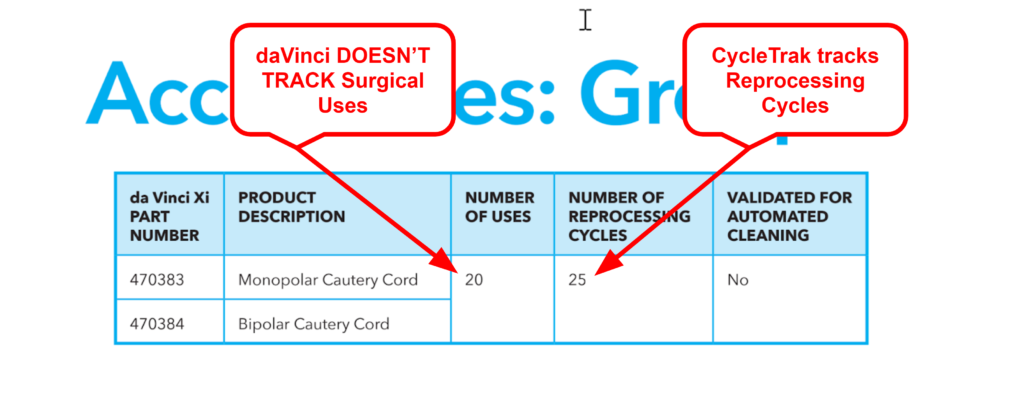
From here — things get less certain. Assumption #3 is true most of the time, but not all of the time. The da Vinci system tracks surgical uses for most of the da Vinci instruments. However, it does not track for cautery cords (two in the Xi line; one in the SP line). Cautery cords have specified surgical uses and reprocessing cycles in the IFU, but the robot does not track surgical uses on these accessories (the cords are not “attached” to the robot). Because the robot doesn’t track surgical uses on these accessories, there is no “remove from service” alert when the maximum threshold has been reached. Therefore, the questions now become: “How does one know when to remove cautery cords from service for exceeding surgical-use or reprocessing cycle thresholds?” “Does one continue to use the cords until they fail and before replacing them?”
Assumption 4: The Most Problematic
The key to the “peel-packing” approach is to assure a process that guarantees surgical uses and reprocessing cycles are always equal. This “always equal” assumption is required for surgical uses to be an indirect tracker or proxy for reprocessing cycles, however, the day-to-day implementation is built upon the promise that the da Vinci instrument is only reprocessed when it has been used in a procedure (i.e. a surgical-use).
This sounds good in theory, but is it realistic?

Or, is it contradictory to the daily reality of operations in an O.R. or a Sterile Processing department? The daily reality may be that an instrument peel pouch was opened in the O.R., but the instrument was not used in the procedure. Perhaps the daily reality may be that an instrument pouch was torn or otherwise compromised. Both of these situations require a reprocessing cycle without a corresponding surgical use.
As a result, surgical uses cannot (and should not) be considered a valid proxy for reprocessing cycles, because they are NOT always equal. Therefore, using surgical uses to track reprocessing cycles for IFU compliance is ultimately a risky proposition.
In conclusion, while “peel-packing” your da Vinci instruments in an effort to not exceed the IFU limit for reprocessing cycles is certainly better than doing nothing, the question becomes: “Is it truly good enough?”
CycleTrak: Your IFU Compliance Solution
CycleTrak is a single, easy solution that eliminates each of the above risks while guaranteeing da Vinci IFU compliance. By simply scanning the unique instrument barcode before it is sterilized, CycleTrak allows efficient and direct tracking of reprocessing cycles on all da Vinci instruments and accessories (cords included).
To learn more about CycleTrak, visit https://www.instrusafe.com/product/cycletrak/.
To schedule a free demo, visit https://www.mycycletrak.com/demo-request.
Author: CycleTrak
